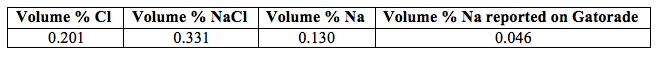Kenzo
The purpose of this experiment was to quantitate the Sodium ions present in a Gatorade Sports Drink through the Mohr Titration Method.
Methods
This experiment was in two phases – (1) the standardization of the 0.1 M AgNO3 and (2) the titration of the Gatorade Sports Drink.
Standardization of 0.1 M AgNO3
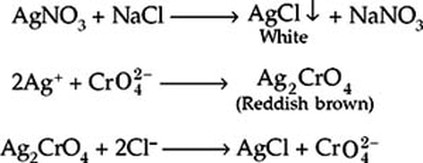
The primary standard used was KCl. This solution was prepared by putting 100mg KCl, 50 mL of ddH20, 3 drops of K2CrO4 solution, and a magnetic stir bar into an Erlenmeyer Flask. The KCl was then titrated with our 0.1 M AgNO3 solution until the appearance of a pale, pink-orange color that remained. The volume of AgNO3 used was then recorded and used to calculate the molarity of the AgNO3 solution.
Titration of Gatorade
The solution was prepared by combining 25ml of the Gatorade and 25ml of ddH20 in an Erlenmeyer Flask and then adding 1ml of potassium chromate indicator. This solution was then titrated with the standardized 0.1 M AgNO3 solution until the appearance of pale red-brown color remained for 30 seconds. The volume of AgNO3 used was then recorded and used to calculate the % Cl and % NaCl in the Gatorade.
This experiment was in two phases – (1) the standardization of the 0.1 M AgNO3 and (2) the titration of the Gatorade Sports Drink.
Standardization of 0.1 M AgNO3
The primary standard used was KCl. This solution was prepared by putting 100mg KCl, 50 mL of ddH20, 3 drops of K2CrO4 solution, and a magnetic stir bar into an Erlenmeyer Flask. The KCl was then titrated with our 0.1 M AgNO3 solution until the appearance of a pale, pink-orange color that remained. The volume of AgNO3 used was then recorded and used to calculate the molarity of the AgNO3 solution.
Titration of Gatorade
The solution was prepared by combining 25ml of the Gatorade and 25ml of ddH20 in an Erlenmeyer Flask and then adding 1ml of potassium chromate indicator. This solution was then titrated with the standardized 0.1 M AgNO3 solution until the appearance of pale red-brown color remained for 30 seconds. The volume of AgNO3 used was then recorded and used to calculate the % Cl and % NaCl in the Gatorade.
Results
The reported percent volume of sodium in the Gatorade volume is smaller compared to the experimentally determined percent volume of sodium. This difference is most likely due to experimental error, not due to the Gatorade bottle with a false report.
The greatest source of experimental error is the titration volume determined through color change of an indicator. This method is less accurate than determining titration with a machine or numerical determination because it is based on the experimenter’s visual acuity in observing a color change. Another source of error in this color change is that the sample of Gatorade had a blue color to it, which will affect the color change expected to be seen.
An improvement to this experiment could be to use colorless Gatorade, as well as standardization of color change observations.