Kenzo
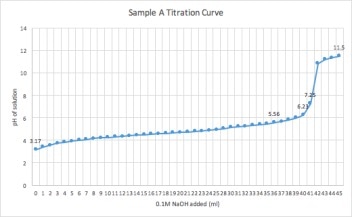
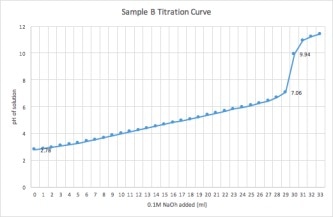
The purpose of this experiment was to perform a simple titration, measure pH using the pH meter, and determine the identity and Titratable Acidity of two unknown samples
Methods
1. 100 mL of sample A was poured into a 250 milliliter beaker. Then 5 drops of both phenolphthalein and methyl orange indicator was added into the sample.
2. The sample was gently stirred on a stirring apparatus, with a stir bar inside.
3.The electrode from the pH meter was submerged into the the sample for continuous recording of the samples pH.
4. 60 mL of 0.1M NaOH was titrated into the sample by increments of 1ml, using an eppendorf multi pipette.
5. This was repeated for sample B.
1. 100 mL of sample A was poured into a 250 milliliter beaker. Then 5 drops of both phenolphthalein and methyl orange indicator was added into the sample.
2. The sample was gently stirred on a stirring apparatus, with a stir bar inside.
3.The electrode from the pH meter was submerged into the the sample for continuous recording of the samples pH.
4. 60 mL of 0.1M NaOH was titrated into the sample by increments of 1ml, using an eppendorf multi pipette.
5. This was repeated for sample B.